Bringing a new MedTech innovation to market is rarely a linear process. Between the first research concept and a fully commercialized product lies a complex development journey where technical feasibility, manufacturability, regulatory requirements, and scalability must all align.
Many promising technologies fail not because the idea lacks potential, but because the transition from R&D to production was not considered early enough. This is where the Technology Readiness Level (TRL) framework becomes valuable — and where Screentec supports customers as a long-term development and manufacturing partner.
At Screentec, we help companies move from concept to scalable manufacturing by combining printed electronics expertise, Design for Manufacturing (DfM), prototyping, pilot production, and ISO 13485-compliant manufacturing under one roof. Our role is not simply to manufacture components, but to help customers reduce technical risk, accelerate commercialization, and create products that are truly ready for real-world use.
What is the Technology Readiness Level (TRL) framework?
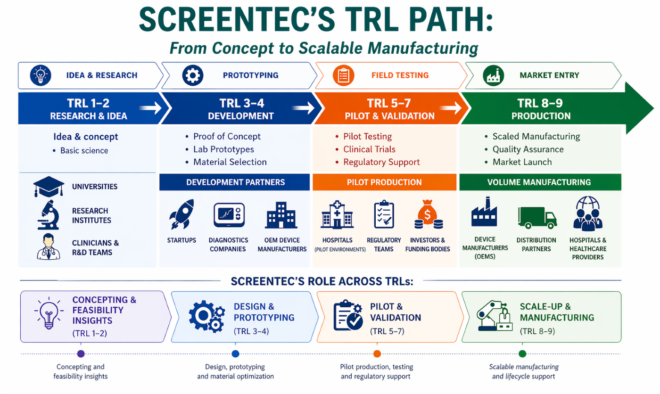
Technology Readiness Levels (TRL 1–9) are used to evaluate how mature a technology is, from early-stage research to commercially validated products. Originally developed by NASA, the framework is now widely used across MedTech, diagnostics, industrial electronics, and wearable device development.
At a simplified level:
TRL 1–2 focuses on research, concepts, and feasibility
TRL 3–4 covers proof of concept and functional prototyping
TRL 5–7 includes validation, pilot production, and industrialization
TRL 8–9 represents validated manufacturing and commercial deployment
For many development teams, the biggest challenge is not generating the initial innovation, it is successfully bridging the gap between prototype development and scalable manufacturing.
Supporting innovation from the earliest stages
At TRL 1–2, the technology exists primarily as a concept. Material choices, substrate compatibility, form factor, and manufacturing method are all still open questions — and the answers to those questions will shape every phase of development that follows.
Screentec engages at this stage by providing feasibility assessments, materials guidance, and application-specific input for printed electronics concepts. Our experience across conductive inks, flexible substrates, electrode structures, and manufacturing processes allows development teams to identify viable paths early and discard approaches that will fail at scale before resources are committed to them.
By considering manufacturability from the start, companies can avoid expensive redesigns later in development.
This early involvement is particularly valuable for university research groups, MedTech startups, OEM R&D teams, and diagnostics developers working to move promising technologies beyond the laboratory. Several of Screentec’s customer relationships have originated from academic research and early-stage innovation environments.
Turning concepts into functional prototypes
At TRL 3–4, the goal shifts from concept validation to building and testing a functional prototype. This is where material selections are made concrete, geometries are defined, and the first real-world performance data is collected.
Screentec supports this phase through optimized design input, material selection, rapid prototyping, and functional validation support across a range of printed electronics applications: including medical electrodes, point-of-care diagnostic components, wearable sensors, and human-machine interface (HMI) systems.
The distinguishing factor in Screentec’s prototyping approach is that every design decision is evaluated against production readiness, not just functional performance. A prototype that performs well but cannot be manufactured consistently is not a development success — it is a problem deferred to a more expensive phase. Screentec’s process prevents that deferral by keeping manufacturability in scope from the first prototype.
Bridging the gap between development and industrialization
A prototype demonstrates that technology can work. A manufacturable product demonstrates that it can work consistently, at volume, within quality and regulatory constraints. Those are fundamentally different challenges, and the distance between them is where most development risk lives.
This is where Screentec provides the greatest value.
At TRL 5–7, Screentec’s focus shifts heavily toward Design for Manufacturing (DfM), process optimization, yield improvement, and pilot production. This phase typically includes:
- Pilot manufacturing runs to validate process repeatability
- Real-environment and integration testing
- Process documentation and validation in support of regulatory submissions
- Yield analysis and design refinement to meet production-grade quality targets
For medical device applications, this work is conducted under Screentec’s ISO 13485-certified quality management system, ensuring that the manufacturing processes being developed meet the traceability and consistency standards required for regulated markets.
What does scalable manufacturing look like at TRL 8–9?
At TRL 8–9, the technology is validated and commercial production becomes the primary focus. For Screentec customers reaching this stage, the transition to volume manufacturing is supported by the same team and processes established during development.
Screentec provides scalable production capabilities supported by robust quality systems, flexible manufacturing, and long-term lifecycle support. For medical applications, our ISO 13485-certified processes help ensure that production meets the strict quality, traceability, and consistency requirements expected in regulated environments.
We support both pilot volumes and larger-scale manufacturing, allowing customers to scale production according to market demand without changing manufacturing partners.
How do you choose the right CDMO partner?
Choosing a development and manufacturing partner for a regulated printed electronics product involves more than evaluating production capabilities. The right partner needs to contribute value across the full TRL path — not just at the manufacturing end.
Key criteria to evaluate:
1.Early-stage engagement: Does the partner bring materials and process expertise to feasibility discussions, or do they only engage once the design is finalized?
2.DfM capability: Is Design for Manufacturing embedded in the development process, or treated as a late-stage check?
3.Regulatory alignment: Are quality systems such as ISO 13485 in place and actively applied during development, not just production?
4.Application range: Can the partner support the specific electrode format, substrate, and gel chemistry your device requires — across wet, dry, and hydrogel formats?
5.Continuity: Will the team that develops your product also manufacture it, preserving institutional knowledge across the product lifecycle?
Screentec is structured to meet all these criteria. By supporting customers across the full Technology Readiness Level path, we help bridge the gap between innovation and industrial production. This integrated approach improves development efficiency, reduces manufacturing risks, and creates a smoother path toward commercialization.
Ready to move your technology forward? Whether your project is at the concept stage or approaching commercial production, contact Screentec to discuss how we can support the next step in your TRL journey.
