
ISO 13485 Certified Medical Device Contract Manufacturing
As an ISO 13485 -certified contract manufacturer, we provide reliable and compliant manufacturing of medical devices and their components.
Screentec Oy has defined and documented the key processes within its quality management system, including their ownership, inputs, and outputs. The core processes cover the entire value chain – from sales and marketing to design, productionization, and final product delivery. This ensures a smooth transition from idea to scalable manufacturing.
Support processes ensure reliable and continuously improving production by maintaining equipment, managing resources, and providing ongoing personnel training.
Our management processes focus on planning, controlling, and evaluating operations to enhance the quality management system (QMS) and ensure compliance with applicable regulatory requirements.
Screentec manufactures medical devices, point-of-care diagnostic solutions, and their components in compliance with ISO 13485 and ISO 9001 standards. We are continuously expanding our capabilities to support customers in regulated industries, with a strong focus on medical device manufacturing and scalable production.
The certification process was conducted by Kiwa Sertifiointi Oy.
You can download our ISO 13485 Certificate HERE.
You can download our ISO 9001 Certificate HERE.
Contract Development and Manufacturing Organization (CDMO)
At Screentec, we support our customers throughout the entire product development lifecycle — from the first idea to a fully commercialized device. The design and productionization services apply to medical devices, diagnostic components, and human-machine interfaces. In production, our skilled and quality-focused operators play a vital role in ensuring that every device is reliable, functional, and fully compliant with all applicable requirements.
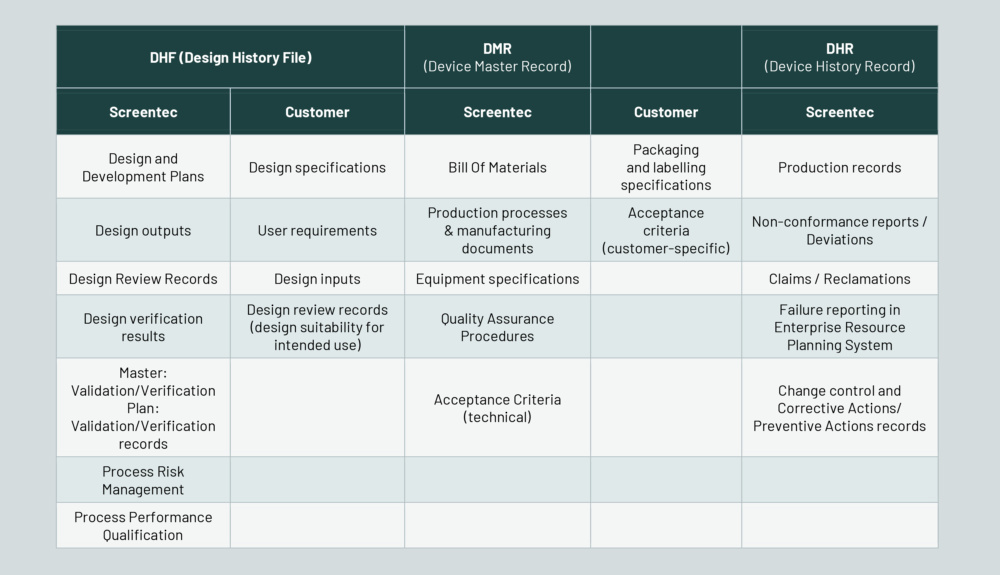 Curious about how our Verification and Validation processes ensure quality and consistency?
Curious about how our Verification and Validation processes ensure quality and consistency?